- Популярные видео
- Авто
- Видео-блоги
- ДТП, аварии
- Для маленьких
- Еда, напитки
- Животные
- Закон и право
- Знаменитости
- Игры
- Искусство
- Комедии
- Красота, мода
- Кулинария, рецепты
- Люди
- Мото
- Музыка
- Мультфильмы
- Наука, технологии
- Новости
- Образование
- Политика
- Праздники
- Приколы
- Природа
- Происшествия
- Путешествия
- Развлечения
- Ржач
- Семья
- Сериалы
- Спорт
- Стиль жизни
- ТВ передачи
- Танцы
- Технологии
- Товары
- Ужасы
- Фильмы
- Шоу-бизнес
- Юмор
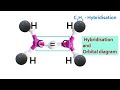
Electronic configuration of LITHIUM(Li) || Condensed electronic configuration of Lithium
This video explains writing electronic configuration of Lithium using s,p,d,f notation, orbital diagram & condensed form.
The arrangement of electrons in the different shells of an atom is called electronic configuration.
To write electronic configuration , calculate the number of electrons from atomic number of an element.
s, p, d & f notation
The number of electrons present in sub shell is depicted as super script & similar sub shell is differentiated by writing the principal quantum number before the sub shell
Lithium( 3) = 1s22s1
orbital diagram
Each orbital is represented as a box and the electron is represented by an arrow.
condensed electronic configuration
We can simplify the electronic configuration of Lithium by representing total number of electrons in the first shell by the name of Helium.The electrons in Helium are called core electrons & the electrons in the highest principal quantum number are called valence electrons.
Видео Electronic configuration of LITHIUM(Li) || Condensed electronic configuration of Lithium канала Aurora Chemistry
The arrangement of electrons in the different shells of an atom is called electronic configuration.
To write electronic configuration , calculate the number of electrons from atomic number of an element.
s, p, d & f notation
The number of electrons present in sub shell is depicted as super script & similar sub shell is differentiated by writing the principal quantum number before the sub shell
Lithium( 3) = 1s22s1
orbital diagram
Each orbital is represented as a box and the electron is represented by an arrow.
condensed electronic configuration
We can simplify the electronic configuration of Lithium by representing total number of electrons in the first shell by the name of Helium.The electrons in Helium are called core electrons & the electrons in the highest principal quantum number are called valence electrons.
Видео Electronic configuration of LITHIUM(Li) || Condensed electronic configuration of Lithium канала Aurora Chemistry
Комментарии отсутствуют
Информация о видео
16 ноября 2025 г. 18:38:59
00:03:55
Другие видео канала