- Популярные видео
- Авто
- Видео-блоги
- ДТП, аварии
- Для маленьких
- Еда, напитки
- Животные
- Закон и право
- Знаменитости
- Игры
- Искусство
- Комедии
- Красота, мода
- Кулинария, рецепты
- Люди
- Мото
- Музыка
- Мультфильмы
- Наука, технологии
- Новости
- Образование
- Политика
- Праздники
- Приколы
- Природа
- Происшествия
- Путешествия
- Развлечения
- Ржач
- Семья
- Сериалы
- Спорт
- Стиль жизни
- ТВ передачи
- Танцы
- Технологии
- Товары
- Ужасы
- Фильмы
- Шоу-бизнес
- Юмор
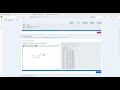
High-potency Fluorenol analogs: –10.7 kcal/mol. Molecular Docking of Selective analogs Dopamine D1A
High-potency Fluorenol analogs: Achieving Binding Energies Exceeding –10.7 kcal/mol. Molecular Docking of Selective analogs Dopamine D1A Receptor.
https://zenodo.org/records/18894579
Keywords: #Fluorenol analogs, #dopaminergic Dopamine D1A Receptor Agonist, Dopamine D2A Receptor, Molecular Docking & Affinity Optimization, Asymmetric Heterocyclic Scaffold, Structure-Activity Relationship (SAR), High-Resolution Binding Energy (–10 kcal/mol), Cyclopentane-fused Imidazoline, Bi-Three-Quadru-Pentadentate Hydrogen Bonding (Fork-like Docking), Conformational Rigidity, Pi-Pi Stacking & Lipophilic Resonance, Molecular Docking
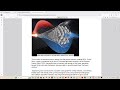
Fluorenol is a selective nootropic agent with a mild dopaminergic effect. It acts as an agonist at the dopamine D1A receptor, forming a hydrogen bond with the Asp103 residue via its hydroxyl group. This interaction occurs without an ionic component (Figure 1), resulting in a calculated binding energy of –8.1 kcal/mol. This, combined with the specific hydrogen bonding of the alcohol, results in the mild pharmacological effect characteristic of this group. Fluorenol shows no significant binding affinity for the D2A receptor, which is a positive characteristic of the fluorene core. However, this agonism, coupled with its relatively low binding energy, requires significant dosages that approach the safety threshold for fused lipophilic aromatic derivatives. Therefore, the search for tautomers, derivatives, and new scaffolds with similar characteristics is a standard tactic. Increasing the binding affinity promotes a more complete direct agonism while minimizing the contribution of reuptake blockade, thus reducing the amount of non-selective dopamine release.
We designed structures with binding energies ranging from –9.0 to –10.7 kcal/mol, which efficiently bind to the Asp103 residue through the hydroxyl group and the nitrogen atoms of 1,2,4-triazole (IIb), 1,2,4-oxadiazole, and imidazole moieties. We thus obtained a gradient of derivatives and analogs, ranging in potency and selectivity. New selectivity data was also obtained, showing that some of the most spatially specific structures for the D1A receptor are inactive at the D2A receptor or are unable to bind to it. While some highly active derivatives are less accessible for organic synthesis than fluorenol, others are equally or even more accessible, maintaining high affinity values and full compliance with the pharmacological profile of fluorenol. The calculated doses of these derivatives and analogs (with binding energies of –9.0 to –10.7 kcal/mol) can vary from several to tens of milligrams, which eliminates issues with accumulation as well as acute and subacute toxicity.
Видео High-potency Fluorenol analogs: –10.7 kcal/mol. Molecular Docking of Selective analogs Dopamine D1A канала Hyper Pharma
https://zenodo.org/records/18894579
Keywords: #Fluorenol analogs, #dopaminergic Dopamine D1A Receptor Agonist, Dopamine D2A Receptor, Molecular Docking & Affinity Optimization, Asymmetric Heterocyclic Scaffold, Structure-Activity Relationship (SAR), High-Resolution Binding Energy (–10 kcal/mol), Cyclopentane-fused Imidazoline, Bi-Three-Quadru-Pentadentate Hydrogen Bonding (Fork-like Docking), Conformational Rigidity, Pi-Pi Stacking & Lipophilic Resonance, Molecular Docking
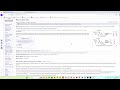
Fluorenol is a selective nootropic agent with a mild dopaminergic effect. It acts as an agonist at the dopamine D1A receptor, forming a hydrogen bond with the Asp103 residue via its hydroxyl group. This interaction occurs without an ionic component (Figure 1), resulting in a calculated binding energy of –8.1 kcal/mol. This, combined with the specific hydrogen bonding of the alcohol, results in the mild pharmacological effect characteristic of this group. Fluorenol shows no significant binding affinity for the D2A receptor, which is a positive characteristic of the fluorene core. However, this agonism, coupled with its relatively low binding energy, requires significant dosages that approach the safety threshold for fused lipophilic aromatic derivatives. Therefore, the search for tautomers, derivatives, and new scaffolds with similar characteristics is a standard tactic. Increasing the binding affinity promotes a more complete direct agonism while minimizing the contribution of reuptake blockade, thus reducing the amount of non-selective dopamine release.
We designed structures with binding energies ranging from –9.0 to –10.7 kcal/mol, which efficiently bind to the Asp103 residue through the hydroxyl group and the nitrogen atoms of 1,2,4-triazole (IIb), 1,2,4-oxadiazole, and imidazole moieties. We thus obtained a gradient of derivatives and analogs, ranging in potency and selectivity. New selectivity data was also obtained, showing that some of the most spatially specific structures for the D1A receptor are inactive at the D2A receptor or are unable to bind to it. While some highly active derivatives are less accessible for organic synthesis than fluorenol, others are equally or even more accessible, maintaining high affinity values and full compliance with the pharmacological profile of fluorenol. The calculated doses of these derivatives and analogs (with binding energies of –9.0 to –10.7 kcal/mol) can vary from several to tens of milligrams, which eliminates issues with accumulation as well as acute and subacute toxicity.
Видео High-potency Fluorenol analogs: –10.7 kcal/mol. Molecular Docking of Selective analogs Dopamine D1A канала Hyper Pharma
Комментарии отсутствуют
Информация о видео
7 марта 2026 г. 1:06:19
00:04:26
Другие видео канала