12. The Shapes of Molecules: VSEPR Theory
MIT 5.111 Principles of Chemical Science, Fall 2014
View the complete course: https://ocw.mit.edu/5-111F14
Instructor: Catherine Drennan
Valence shell electron pair repulsion or VSEPR theory can be used to predict molecular geometry. The theory is based on Lewis structures and the simple idea that that the preferred geometry around a central atom is the one that minimizes electron repulsion. Chemist Stefanie Sydlik tells us how she uses VSEPR theory to design sensors that are capable of detecting landmines.
License: Creative Commons BY-NC-SA
More information at http://ocw.mit.edu/terms
More courses at http://ocw.mit.edu
Видео 12. The Shapes of Molecules: VSEPR Theory канала MIT OpenCourseWare
View the complete course: https://ocw.mit.edu/5-111F14
Instructor: Catherine Drennan
Valence shell electron pair repulsion or VSEPR theory can be used to predict molecular geometry. The theory is based on Lewis structures and the simple idea that that the preferred geometry around a central atom is the one that minimizes electron repulsion. Chemist Stefanie Sydlik tells us how she uses VSEPR theory to design sensors that are capable of detecting landmines.
License: Creative Commons BY-NC-SA
More information at http://ocw.mit.edu/terms
More courses at http://ocw.mit.edu
Видео 12. The Shapes of Molecules: VSEPR Theory канала MIT OpenCourseWare
Показать
Комментарии отсутствуют
Информация о видео
Другие видео канала
 13. Molecular Orbital Theory
13. Molecular Orbital Theory VSEPR Theory: Introduction
VSEPR Theory: Introduction 11. Formal Charge and Resonance
11. Formal Charge and Resonance 8.01x - Lect 27 - Fluid Mechanics, Hydrostatics, Pascal's Principle, Atmosph. Pressure
8.01x - Lect 27 - Fluid Mechanics, Hydrostatics, Pascal's Principle, Atmosph. Pressure Quick Way to Memorize Molecular Geometry | Polarity | Angle | Hybridization | Ace That Exam
Quick Way to Memorize Molecular Geometry | Polarity | Angle | Hybridization | Ace That Exam VSEPR Theory - Balloons
VSEPR Theory - Balloons Introduction to Aerospace Engineering: Aerodynamics
Introduction to Aerospace Engineering: Aerodynamics 15. Thermodynamics: Bond and Reaction Enthalpies
15. Thermodynamics: Bond and Reaction Enthalpies 16. Thermodynamics: Gibbs Free Energy and Entropy
16. Thermodynamics: Gibbs Free Energy and Entropy Introduction to Aerospace Engineering: The Scale of Things
Introduction to Aerospace Engineering: The Scale of Things Valence Shell Electron Pair Repulsion Theory (VSEPR Theory)
Valence Shell Electron Pair Repulsion Theory (VSEPR Theory) 28. Transition Metals: Crystal Field Theory Part I
28. Transition Metals: Crystal Field Theory Part I VSEPR Theory and Molecular Geometry
VSEPR Theory and Molecular Geometry 1. The Importance of Chemical Principles
1. The Importance of Chemical Principles 3. Wave-Particle Duality of Light
3. Wave-Particle Duality of Light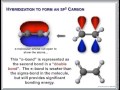 Organic Chemistry McMurry Chapter 1, Structure and Bonding
Organic Chemistry McMurry Chapter 1, Structure and Bonding Lewis Structures, Introduction, Formal Charge, Molecular Geometry, Resonance, Polar or Nonpolar
Lewis Structures, Introduction, Formal Charge, Molecular Geometry, Resonance, Polar or Nonpolar 4. Wave-Particle Duality of Matter; Schrödinger Equation
4. Wave-Particle Duality of Matter; Schrödinger Equation VSEPR and Molecular Geometry: Rules, Examples, and Practice
VSEPR and Molecular Geometry: Rules, Examples, and Practice 6. Hydrogen Atom Wavefunctions (Orbitals)
6. Hydrogen Atom Wavefunctions (Orbitals)