Practice with Redox Spontaneity and Standard Cell Potential | OpenStax Chemistry 2e 17.3
00:00 Finding the Half-reactions
02:41 The Full Redox Equation
03:47 Laying out the Reduction Potentials
05:03 The Hypothetical Galvanic Cell
06:42 Spontaneity and Cell Potential
Видео Practice with Redox Spontaneity and Standard Cell Potential | OpenStax Chemistry 2e 17.3 канала Michael Evans
02:41 The Full Redox Equation
03:47 Laying out the Reduction Potentials
05:03 The Hypothetical Galvanic Cell
06:42 Spontaneity and Cell Potential
Видео Practice with Redox Spontaneity and Standard Cell Potential | OpenStax Chemistry 2e 17.3 канала Michael Evans
Показать
Комментарии отсутствуют
Информация о видео
Другие видео канала
 12.01 Introduction to Aldol Additions
12.01 Introduction to Aldol Additions Structure and Nomenclature of Polymers
Structure and Nomenclature of Polymers Determining Molecular Geometry | Practice Exam 3.1 | Fall 2021
Determining Molecular Geometry | Practice Exam 3.1 | Fall 2021 Electron Configurations of Transition Metals | OpenStax Chemistry 2e 19.1
Electron Configurations of Transition Metals | OpenStax Chemistry 2e 19.1 Stereochemistry of Unimolecular Nucleophilic Substitutions
Stereochemistry of Unimolecular Nucleophilic Substitutions Introducing Carboxylic Acids; Acidity of Carboxylic Acids
Introducing Carboxylic Acids; Acidity of Carboxylic Acids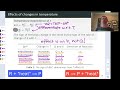 Temperature, Catalysts, and Shifting Equilibria | OpenStax Chemistry 2e 13.3
Temperature, Catalysts, and Shifting Equilibria | OpenStax Chemistry 2e 13.3 5.5 Electron rich Sensitizers: Patterns in Structure and Reactivity
5.5 Electron rich Sensitizers: Patterns in Structure and Reactivity Regioselectivity in Electrophilic Aromatic Substitutions
Regioselectivity in Electrophilic Aromatic Substitutions Review of Chemical Thermodynamics for Organic Chemistry
Review of Chemical Thermodynamics for Organic Chemistry Introduction to Buffers | OpenStax Chemistry 2e 14.6
Introduction to Buffers | OpenStax Chemistry 2e 14.6 Functional Groups
Functional Groups 7.2 Analysis of Sigma-bond Cleavage from an Excited State
7.2 Analysis of Sigma-bond Cleavage from an Excited State Determining Isomeric Relationships
Determining Isomeric Relationships 14.04 The Michael Reaction
14.04 The Michael Reaction Introduction to Organic Synthesis
Introduction to Organic Synthesis 1.6 Length, Time, and Energy Scales of Photochemistry
1.6 Length, Time, and Energy Scales of Photochemistry 2.2 Excitation as Electron Promotion
2.2 Excitation as Electron Promotion Application of General Acid Catalysis
Application of General Acid Catalysis 11.05 General Reactivity of Enols and Enolates
11.05 General Reactivity of Enols and Enolates Electrolysis and Stoichiometry | OpenStax Chemistry 2e 17.7
Electrolysis and Stoichiometry | OpenStax Chemistry 2e 17.7